-
中文名称:三氯乙烯
-
英文名称:Trichloroethylene
-
CAS No.:79-01-6
-
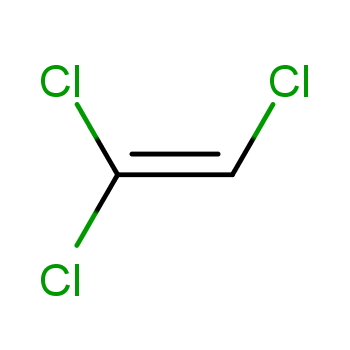
分子式:C2HCl3
-
分子量:131.38
-
EINECS号:201-167-4
-
中文别名
三氯代乙烯无水三氯乙烯1,1,2-三氯乙烯
-
英文别名
Trichloroethylene, StabilizedTrichloroethylene, SuperDry, stabilized, J&KSealMFCD00000838TRICHLOROETHYLENE EMPLURA 1 LTRICHLOROETHYLENE EMPLURA 25 LTRICHLOROETHYLENE EMPLURA 190 LTRICHLOROETHYLENE FOR ANALYSIS EMSUREEINECS 201-167-4Trichloroethylene, stabilized with 400 ppM triethylaMineTrichloroethylene, Stabilized, SpectroSolv (TM)
-
CAS.79-01-6
Inchi
InChI=1S/C2HCl3/c3-1-2(4)5/h1H
InChIkey
XSTXAVWGXDQKEL-UHFFFAOYSA-N
Canonical Smiles
C(=C(Cl)Cl)Cl
国际化联命名
1,1,2-trichloroethene
-
密度:1.462
-
熔点:-86℃
-
沸点:-99° F (NTP, 1992)
-
折射率:1.476-1.478
-
闪光点:greater than 200° F (NTP, 1992)
-
蒸汽压:72.4±0.1 mmHg at 25°C
-
PSA:0.00000
-
logP:2.50170
-
溶解度: 0.11 g/100 mL
-
黏性:Viscosity is a measure of a fluid's resistance to flow. It describes the internal friction of a moving fluid.
-
蒸汽密度:4.5 (vs air)
-
分析实验室方法:Method: NIOSH 1022, Issue 2; Procedure: gas chromatography with flame ionization detector; Analyte: trichloroethylene;; Matrix: air; Detection Limit: 0.01 mg/sample.
-
外观:无色透明液体,有似氯仿的气味。
-
大气OH速率常数:2.36e-12 cm3/molecule*sec
-
自燃:770° F (USCG, 1999)
-
存储:储存注意事项 储存于阴凉、通风的库房。远离火种、热源。库温不超过32℃,相对湿度不超过80%。包装要求密封,不可与空气接触。应与氧化剂、还原剂、碱类、金属粉末、食用化学品分开存放,切忌混储。不宜大量储存或久存。配备相应品种和数量的消防器材。储区应备有泄漏应急处理设备和合适的收容材料。
-
自燃温度:770 °F (USCG, 1999)
-
致癌性:Trichloroethylene is reasonably anticipated to be a human carcinogen based on limited evidence of carcinogenicity from studies in humans, sufficient evidence of carcinogenicity from studies in experimental animals, and information from studies on mechanisms of carcinogenesis.
-
化学性质:Trichloroethylene (TCE) is a clear, colorless, nonflammable (at room temperature) stable toxic liquid with chloroform-like odor (ATSDR, 2011). It is slightly soluble in water, is soluble in greases and common organic solvents, and boils at 87°C (190 F).On contact with air, it slowly decomposes and forms phosgene, hydrogen chloride, and dichloroacetyl chloride. Trichloroethylene in contact with water becomes corrosive and forms dichloroacetic acid and hydrochloric acid. It is soluble in methanol, diethyl ether, and acetone.
-
物理属性:Clear, colorless, watery-liquid with a chloroform-like odor. Odor threshold concentrationsdetermined in air were 21.4 ppmv (Leonardos et al., 1969) and 3.9 ppmv (Nagata and Takeuchi,1990). The average least detectable odor threshold concentrations in water at 60 °C and in air at 40°C were 10 and 2.6 mg/L, respectively (Alexander et al., 1982).
-
颜色:无色透明液体,有似氯仿的气味。[1]
-
接触性:Trichloroethylene is a chlorinated hydrocarbon used asa detergent or solvent for metals, oils, resins, sulfur, andas general degreasing agent. It can cause irritant contactdermatitis, generalized exanthema, Stevens-Johnson-like syndrome, pustular or bullous eruption, scleroderma,as well as neurological and hepatic disorders.
-
腐蚀性:Non-corrosive
-
分解:Hazardous decomposition products formed under fire conditions - Carbon oxides, hydrogen chloride; gas.
-
燃烧热:-6.56 kJ/g
-
汽化热:34.54 kJ/mol at 25 deg C; 31.40 kJ/mol at 87.21 deg C
-
亨利法律常量:0.01 atm-m3/mole
-
电离电位:9.45 eV
-
气味:Chloroform-like; ethereal.
-
气味阈值:3.9ppm
-
物理属性:三氯乙烯(79-01-6 )性状: (1)沸点86.7℃,相对密度(125612) 1.456~1.462,闪点32.2℃,熔点-73℃,折射率(n 2020)1.477。 (2)澄清无色流体,具有氯仿似甜的气味。 (3)微溶于水,混溶于乙醇、丙酮、乙醚、四氯化碳。 (4)性能稳定,即使在湿空气中,也不腐蚀普通金属。质量指标 FAO/WHO,1992 (暂定) 1.馏程 85~88℃ 2.不挥发残渣(OT-39) ≤20mg/kg 3.氯化物试验 阴性 4.炔属化合物试验 阴性 5.酸度试验 阴性 6.游离氯试验 阴性 7.碳酰氯试验 阴性毒性: LD50 3670mg/kg(大鼠,经口)。 无色稳定、低佛点重质油状液体,具有类似氯仿的气味。 与一般有机溶剂混溶,微溶于水。 性状 无色稳定、低佛点重质油状液体,具有类似氯仿的气味。 熔点 -87.1℃ 沸点 86.7℃ 相对密度 1.456~1.462 折射率 1.4782 闪点 32.2℃ 溶解性 与一般有机溶剂混溶,微溶于水。
-
溶解性:不溶于水,溶于乙醇、乙醚,可混溶于多数有机溶剂。[15]
-
光谱特性:SADTLER REF NUMBER: 185 (IR, PRISM); MAX ABSORPTION: LESS THAN 200 NM (VAPOR)
Index of refraction: 1.4773 @ 20 deg C/D
IR: 62 (Sadtler Research Laboratories IR Grating Collection)
NMR: 9266 (Sadtler Research Laboratories Spectral Collection)
MASS: 583 (Atlas of Mass Spectral Data, John Wiley & Sons, New York)
Intense mass spectral peaks: 60 m/z, 95 m/z, 130 m/z
-
稳定性:Stable. Incompatible with oxidizing agents, aluminium, magnesium, strong bases, reducing agents. Light-sensitive. Reacts violently with many metals, ozone, potassium nitrate, potassium hydroxide, sodium hydroxide.
-
存储温度:0-6°C
-
表面拉伸:0.0264 N/m at 20 deg C
-
毒性摘要:IDENTIFICATION AND USE: Trichloroethylene; (TCE) is a colorless liquid (unless dyed blue). The major use of TCE is in metal cleaning or degreasing. TCE was used earlier as an extraction solvent for natural fats and oils, such as palm, coconut and soya bean oils. It was also an extraction solvent for spices, hops and the decaffeination of coffee. The United States Food and Drug Administration banned these uses of trichloroethylene;. Its use in cosmetic and drug products was also discontinued. It was also used as both an anesthetic and an analgesic in obstetrics. HUMAN EXPOSURE AND TOXICITY: Potential symptoms of overexposure are headache, vertigo, visual disturbance, fatigue, giddiness, tremors, somnolence, nausea and vomiting, irritation of eyes and skin, dermatitis, cardiac arrhythmias, paresthesia, liver injury. Death has occurred at very high concentrations (10,000 ppm) and was associated with cardiac arrhythmia and massive liver damage. Workers chronically exposed to levels between 38 and 172 ppm reported symptoms of sleepiness, dizziness, headache, and nausea, but no apparent trigeminal nerve disorders. In a study of Dutch workers regularly exposed to no more than 35 ppm, investigators found no trigeminal nerve impairment as measured by blink reflex, but did observe a significant association between years of exposure and masseter reflex, which is another measure of trigeminal nerve function. Increased micronucleus frequency is associated with occupational TCE exposure. TCE exerts genotoxic effects in HepG2 cells. In Tier I cancer incidence cohort studies, TCE exposure was associated with an increased risk of kidney cancer. Liver cancer incidence was elevated in most of the Tier I cancer incidence studies. Maternal residential proximity to industrial emissions of chlorinated solvents might be associated with selected birth defects in offspring, especially among older mothers. ANIMAL STUDIES: Studies on the longer-term toxicity of TCE in rats and mice exposed orally and by inhalation showed consistent increases in relative liver weight and associated histopathological and biochemical changes. The effects described in kidney included increased relative weights in mice exposed continuously to > 75 ppm (> 390 mg/cu m) TCE for 30 days and renal dysfunction in the absence of marked histopathological changes in rats exposed to > 50 ppm (> 260 mg/cu m) for 12 weeks. In rats exposed to TCE by gavage (50 or 250 mg/kg, once daily, 4 to 5 days/week for 52 weeks) there was a dose-related increase in the incidence of leukemia (immunoblastic lymphosarcomas) in males. No increase was noted in the tumor incidence of females. In TCE exposed rat and mice (7 hours/day, 5 days/week for 104 weeks at 50, 150, or 450 ppm), tumors were found mainly in the hematopoietic system, lungs, and mammary glands of mice and in the pituitary and mammary glands of rats. Administration of TCE in the diet of mice and rats at concentrations equivalent to doses of up to 300 mg/kg bw per day for two generations resulted in marginal effects on testicular weight and on survival of pups of both the F1 and F2 generations at the highest dose. In general, TCE and most of its major metabolites are not potent genotoxicants in a broad range of bacterial, lower eukaryotic, and in vitro and in vivo mammalian test systems. In mammalian cell-culture studies, TCE did not induce chromosomal aberrations in Chinese hamster ovary (CHO) cells, unscheduled DNA synthesis in rat hepatocytes, but it did induce sister chromatid exchange in CHO cells, gene mutations in mouse lymphoma cells, and morphological transformation of rat embryo cells. In rodent in vivo studies, TCE did not induce unscheduled DNA synthesis, sister chromatid exchange, dominant lethal mutations, or chromosomal aberrations. TCE gave mixed results for DNA single-strand breaks or alkali-labile sites in mouse liver and positive results for micronucleus formation in mice. ECOTOXICITY STUDIES: TCE had effects on genes and proteins related to metabolism, reproduction, and growth in D. magna. Exposure of goldfish (Carassius auratus) to 0.1 mg/L TCE for >/= 60 days in a static-renewal test resulted in significantly reduced body weight and altered histopathology. Affected fathead minnows, 31 days old, in toxicant concentrations ranging from 8.43-77.3 mg/L, lost schooling behavior, swam in a corkscrew/spiral pattern near the surface, were hyperactive and hemorrhaging. TCE induced chlorosis (bleaching of needles), necrosis (death of needles), and premature needle loss over 2 decades in fir (Abies alba), Norway spruce (Picea abies), beech (Fagus silvatica), and other tree species.
-
用途 用作不燃性溶剂和分析试剂;
用于制造靛蓝及其他染料,生产一氯代乙酸,是重要的工业溶剂,用作金属洗涤剂、干洗剂、农用杀虫剂等;
萃取溶剂。主要用于咖啡的脱咖啡因等,其在磨粒咖啡成品中的 允许残留量为25mg/kg,在速溶咖啡中为10mg/kg,在香辛料油树脂中为30mg/kg。;
优良的溶剂,用作金属表面处理剂,电镀、上漆前的清洁剂,金属脱脂剂和脂肪、油、石蜡的萃取剂。用于有机合成、农药的生产。三氯乙烯 用于生产四氯乙烯可作为驱肠虫药;用于生产六氯乙烷,作为兽用驱虫药,来用防治反刍兽类肝蛭病及胃蛭病、睾吸虫病和线虫病。;
用作不燃性溶剂、测定碘价、有机合成。;